Are you skeptical of what you read?
There is an on-line community called HASTRO-L, of historians of astronomy and people with an interest in the history of astronomy. It grew out of the University of Notre Dame’s regular history of astronomy conferences. There is a person who hangs out on HASTRO-L who I would describe as being very skeptical of what he reads and rejecting a lot of the ideas of modern astronomy. He has developed his own cosmology, based on the ideas of the sixteenth-century astronomer Tycho Brahe. I know what you are thinking—“another crank who claims that modern science is wrong”—but bear with me. Sometimes the arguments of such folks are pretty intriguing, and can be educational.
This fellow, Simon Shack, calls his cosmology the TYCHOS system. He has sunk much time and effort into a website advocating for his system. Last year, he directed the attention of HASTRO-L to a post on that website (click here for it) where he noted that, according to Wikipedia, the angular diameter of the star Promixa Centauri as seen from Earth is 0.001 seconds of arc (0.001´´).
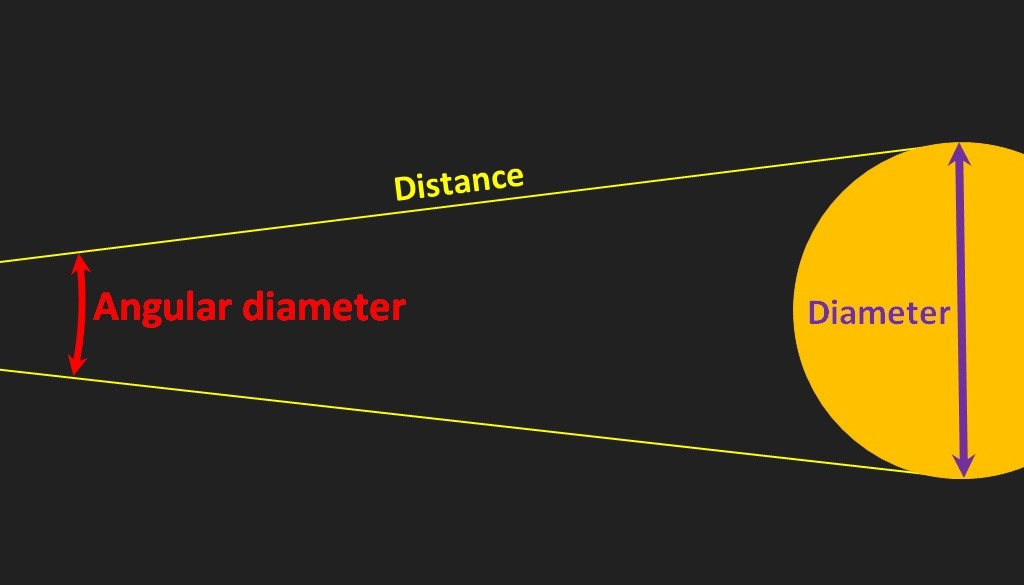
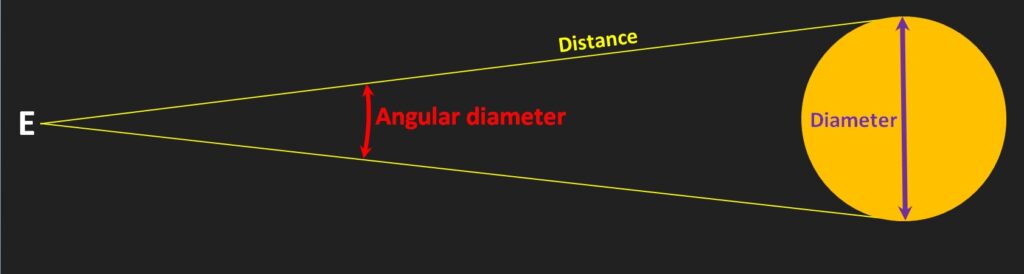
The angular diameter of an object depends on its diameter and its distance. The farther away an object is, or the smaller the object is, the smaller is its angular diameter (as seen by the eye at E, below). There are 60 seconds in an arc minute and sixty minutes in a degree, so this value for Proxima is 1/1000th of 1/60th of 1/60th of a degree. It’s small.
Shack continues:
Wow… [this is] almost 2 MILLION TIMES smaller than the angular diameter subtended by the Sun in our skies! Now, as we go along, please keep in mind that we are talking about the VERY NEARMOST STAR to our Solar System, ok?…
HOW – pray tell – would it possibly be visible from the Earth at all? Can your mind even remotely conceptualize such a possibility?
He is right! And there is much to be learned here.
Proxima is not the best choice. It is not visible to the naked eye. Let’s choose stars we can see with our own two eyes—stars that are truly visible from Earth. After all, if we are going to be science skeptics, let’s vehemently be science skeptics.
Consider Deneb and Sirius instead. These are certainly “visible”, as in “seeable by our unaided human vision”, from Earth. Wikipedia says Deneb has an angular diameter of 0.002´´, while Sirius has an angular diameter of 0.006´´. (I checked the Wikipedia numbers. They are good.)
Modern astronomy tells us that these tiny values are because Deneb and Sirius are sun-like bodies at vast, vast distances. The angular diameter of the Sun is about 0.5 degrees (0.5°), or 1800´´. The Sun is 93,000,000 miles or 149,600,000 km distant. If we pushed the Sun out to a distance of 10 light-years (a little past Sirius), what would its angular diameter be? One light-year is 9,460,000,000,000 km, so 10 lyr = 94,600,000,000,000 km. Pushing the Sun out to 10 lyr is pushing it so that it is 94,600,000,000,000 km / 149,600,000 km = 632,353 times farther away. That would make the Sun’s angular diameter 632,353 times smaller, or 1800´´ / 632,353 = 0.003´´ (obviously Sirius is a somewhat bigger star than the Sun).
But let’s compare Deneb and Sirius to the Moon, rather than the Sun. Both Moon and Sun have about the same angular diameter. Since we see both Moon and stars together in the sky at the same time, the Moon seems a better object of comparison.
The Moon is 1800´´ / .002´´ = 900,000 times the angular diameter of Deneb. Area goes as the diameter squared. Therefore the Moon occupies 900,000 x 900,000 = 810,000,000,000 (that is 0.81 trillion) times the space in the sky as Deneb.
The Moon is not so big, O Readers! Find a small round object that measures 4 or 5 mm in diameter, like a pea. Hold it about 50 cm, or 20 inches—roughly arm’s length—from your eye. The angular diameter of that pea is about 0.5°. If you do not believe it (be skeptical, after all), take it outside when the Moon is out and see for yourself.
So, what size object is 1/900,000th the diameter of a pea? 4.5 mm is 0.45 cm. 0.45 cm / 900,000 = 0.0000005 cm.
How small is 0.0000005 cm? It’s small! An atom is roughly 0.00000001 cm in diameter (a carbon atom is a little smaller, at about 0.000000007 cm; a lead atom is a bit bigger, at about 0.000000018 cm). So, in terms of atoms, our object-that-is-1/900,000th-of-the-diameter-of-a-pea is 0.0000005 cm/0.00000001 cm = 50 atoms in diameter!
So seeing Deneb is like seeing a ball of atoms, only 50 atoms in diameter, held 50 cm from your eye.
Are you skeptical now? Does the TYCHOS advocate have a point?
You cannot see 50 atoms in row. You cannot see a ball of atoms, 50 wide. And you certainly cannot see that ball as well as you can see Deneb! Sirius would be like seeing a ball that is 150 atoms wide—still not possible.
Shack says this “demonstrates the utter absurdity” of modern ideas about stars and their distances. Does it? After all, we truly cannot see such small things. Therefore, mustn’t it be the case that the stars cannot have such small angular diameters? Mustn’t astronomers be wrong? Logic dictates that the angular diameters of stars must be much, much larger, since we can see them. That in turn would mean that the stars either have to be much, much closer than astronomers say, or they have to be much, much bigger than the sun. Shack’s argument is excellent! It is exactly the type of argument that was in play around in Brahe’s time.
People like Johannes Kepler argued back then that the stars were very, very large. He believed Copernicus was right, but that the Sun was the only Sun in the universe—the rest of the universe was just these vast stars in space. He was very opposed to ideas like those of Giordano Bruno who said that stars were other suns. People like Brahe, Kepler’s boss, argued back then that the stars were not so far away. That meant Copernicus was wrong, because the motion of the Earth would be noticeably reflected in close-by stars. Brahe argued that Earth was at rest.
But it turns out that, while you cannot see a 50-atom-wide ball at arm’s length, you can see the light from it (more on seeing atoms in a moment). You can see the light from a bright object, even if you cannot see the object itself. To test this (be skeptical!), you need a bright, but very small, light. The single, bright LED on a smart phone’s “flashlight” feature is good for this.
In the daylight, set the phone up somewhere, with the LED off. Walk away from it until you can barely see the phone itself, not to mention the LED. Mark that spot—“X”!
Now, when it is dark, set the phone up in the same place. Turn on the LED and aim it straight at “X”. Then go to “X” and look back at the phone. You could not see the dark LED in the daylight, but you will be able to see its light in the dark! Indeed, the glow of the LED will probably look larger than the phone itself looked in the daylight.
That is why we can see the stars—or, more correctly, why we can see their light. We can’t actually see them. Like Shack says, they are too small.
The physics of light that undergirds all this did not get worked out until long after Brahe and Kepler. Thus it took a long time for astronomers to really understand what they were looking at when they looked at stars—a lot longer than what you read in the history books.
After first writing this post, I learned of a purported photograph of a single atom, taken in 2020:
An image of a single positively-charged strontium atom, held near motionless by electric fields, has won the overall prize in a national science photography competition, organised by the [UK’s] Engineering and Physical Sciences Research Council (EPSRC).
‘Single Atom in an Ion Trap’, by David Nadlinger, from the University of Oxford, shows the atom held by the fields emanating from the metal electrodes surrounding it. The distance between the small needle tips is about two millimetres.
When illuminated by a laser of the right blue-violet colour the atom absorbs and re-emits light particles sufficiently quickly for an ordinary camera to capture it in a long exposure photograph. The winning picture was taken through a window of the ultra-high vacuum chamber that houses the ion trap.
Here is the image (the atom is indicated by the arrow):

And here is Nadlinger explaining how the photograph came about:
The idea of being able to see a single atom with the naked eye had struck me as a wonderfully direct and visceral bridge between the miniscule quantum world and our macroscopic reality. A back-of-the-envelope calculation showed the numbers to be on my side, and when I set off to the lab with camera and tripods one quiet Sunday afternoon, I was rewarded with this particular picture of a small, pale blue dot.
But are we seeing a single atom here? Let us look closely. The distance between the needle tips is 2 mm. The “atom” is small, but its width is a measurable fraction of that 2 mm gap.

If I copy and paste the “atom” at regular intervals, I get the picture below. There are 7 “atoms” visible now, and I could certainly fit another “atom” in each gap between “atoms”. That would be 15 “atoms” from needle to needle. But I think I could fit a little more in there, so let’s say I could fit 20 “atoms” between the needle tips in the picture. With a 2 mm gap and 20 to fit the gap, that makes our “atom” 2 mm / 20 = 0.1 mm = 0.01 cm wide.

That is huge! Human hairs and such are smaller than that, and we can see them with our naked eyes. Atoms are incomparably smaller. Recall that a lead atom was 0.000000018 cm—a million times smaller. We are not seeing a strontium atom here—we are only seeing its light. Just like with the stars.